Calcium »
PDB 6oly-6p8x »
6op4 »
Calcium in PDB 6op4: Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii
Enzymatic activity of Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii
All present enzymatic activity of Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii:
1.18.6.1;
1.18.6.1;
Protein crystallography data
The structure of Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii, PDB code: 6op4
was solved by
R.J.Arias,
D.C.Rees,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 39.41 / 2.30 |
| Space group | P 1 21 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 76.981, 129.326, 106.755, 90.00, 108.84, 90.00 |
| R / Rfree (%) | 17.6 / 22.3 |
Other elements in 6op4:
The structure of Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii also contains other interesting chemical elements:
| Molybdenum | (Mo) | 2 atoms |
| Magnesium | (Mg) | 2 atoms |
| Iron | (Fe) | 30 atoms |
Calcium Binding Sites:
The binding sites of Calcium atom in the Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii
(pdb code 6op4). This binding sites where shown within
5.0 Angstroms radius around Calcium atom.
In total 2 binding sites of Calcium where determined in the Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii, PDB code: 6op4:
Jump to Calcium binding site number: 1; 2;
In total 2 binding sites of Calcium where determined in the Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii, PDB code: 6op4:
Jump to Calcium binding site number: 1; 2;
Calcium binding site 1 out of 2 in 6op4
Go back to
Calcium binding site 1 out
of 2 in the Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii
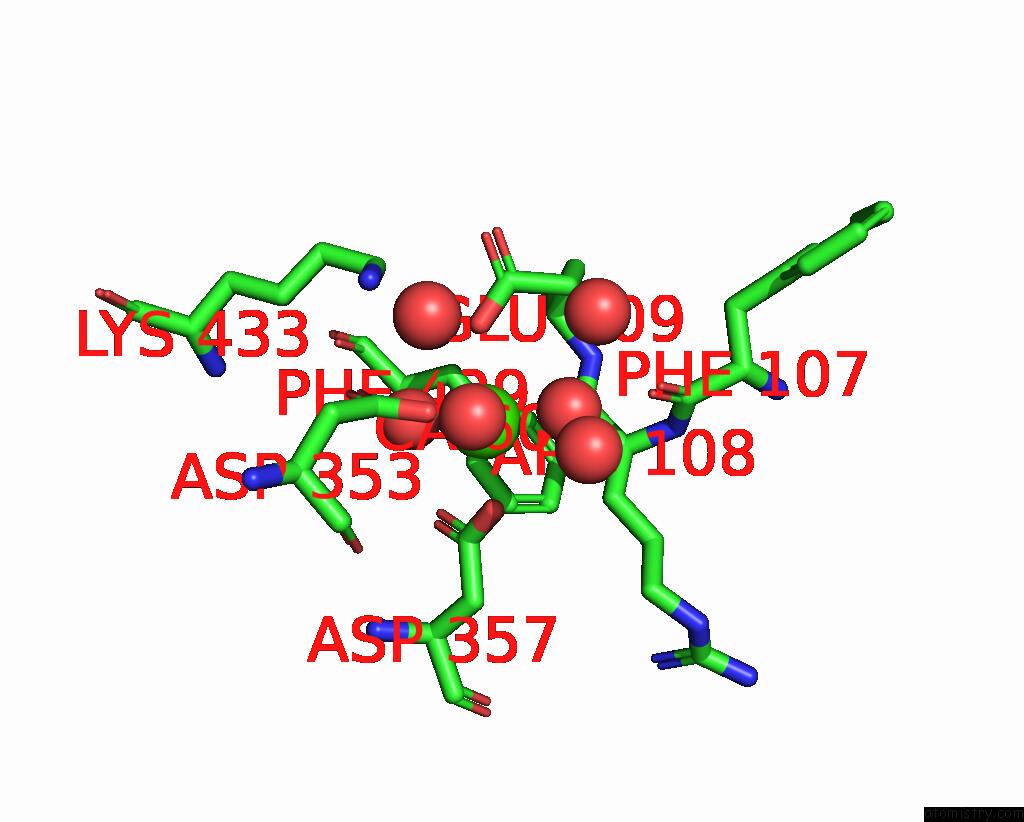
Mono view
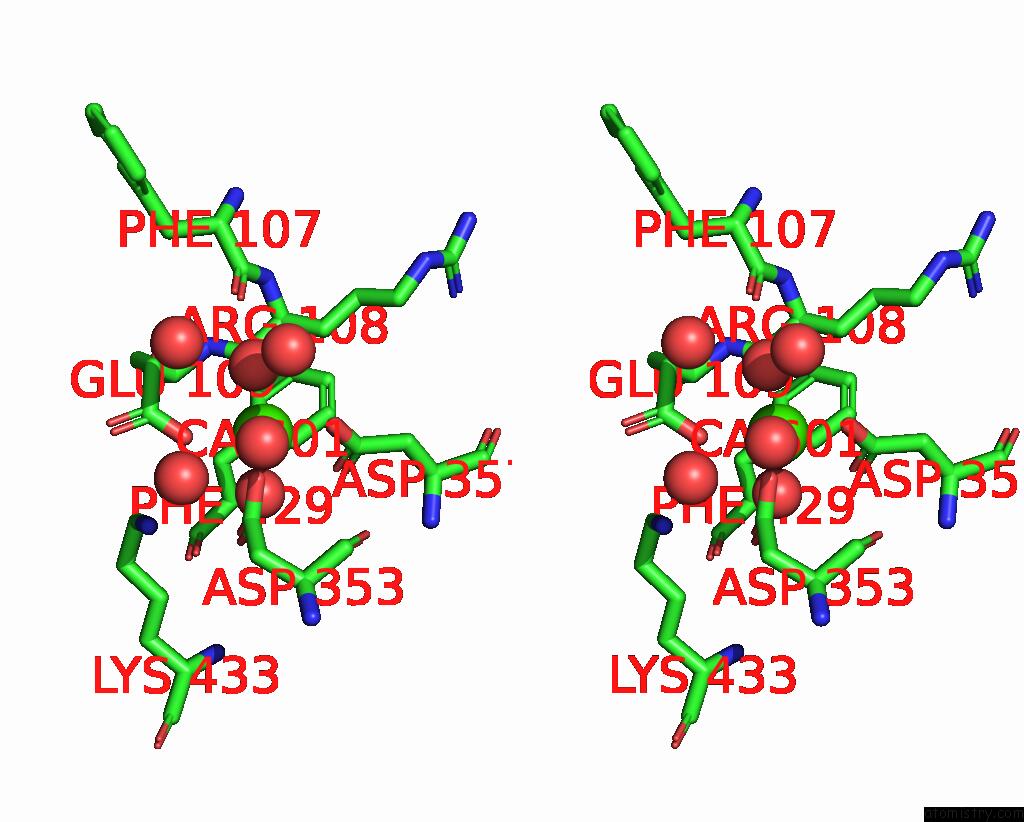
Stereo pair view

Mono view

Stereo pair view
A full contact list of Calcium with other atoms in the Ca binding
site number 1 of Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii within 5.0Å range:
|
Calcium binding site 2 out of 2 in 6op4
Go back to
Calcium binding site 2 out
of 2 in the Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii
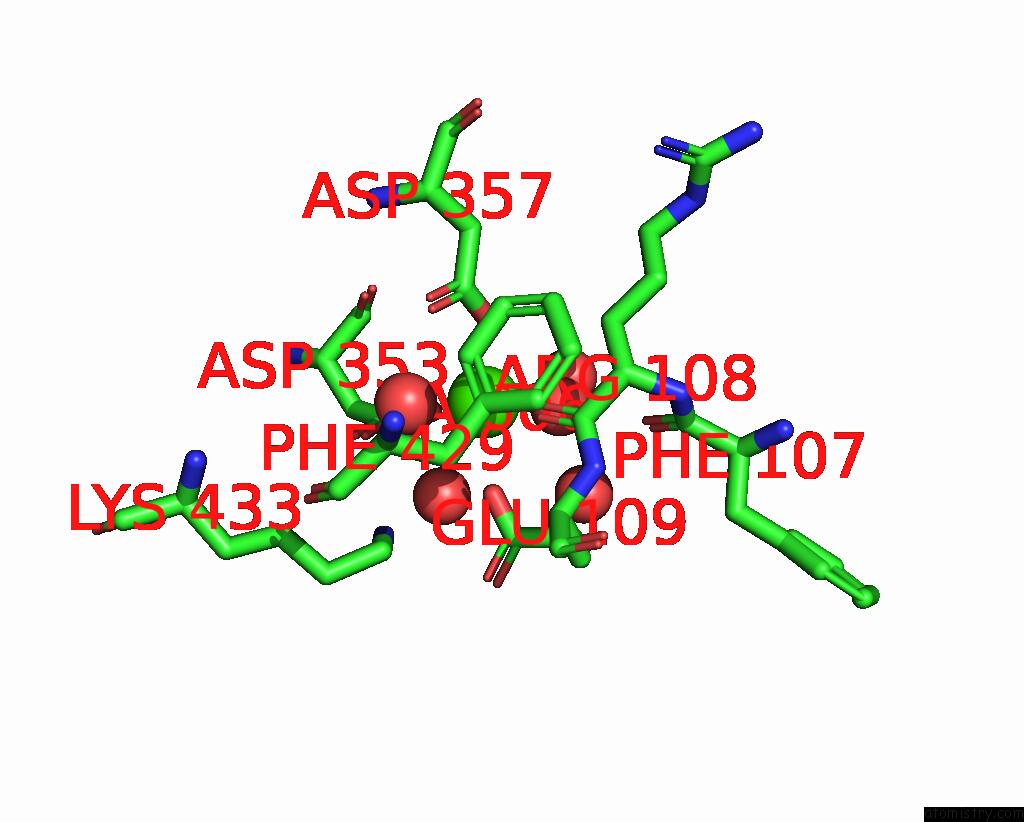
Mono view
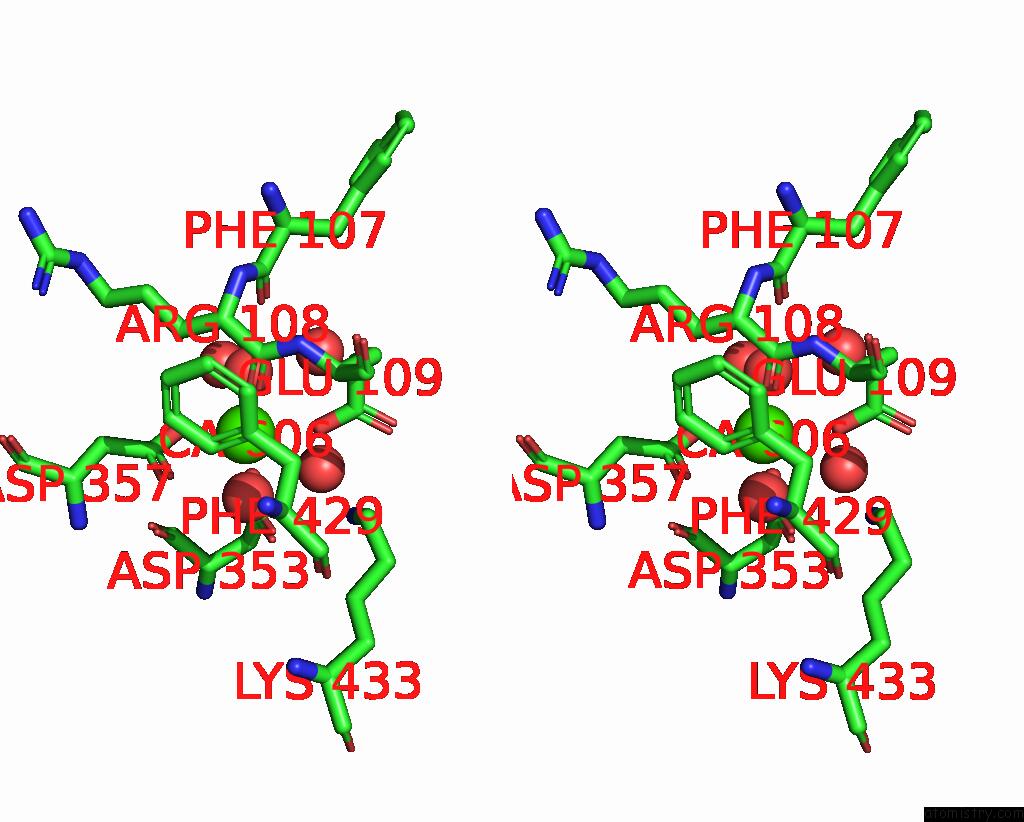
Stereo pair view

Mono view

Stereo pair view
A full contact list of Calcium with other atoms in the Ca binding
site number 2 of Selenium-Incorporated, Carbon Monoxide-Inhibited, Reactivated Femo- Cofactor of Nitrogenase From Azotobacter Vinelandii within 5.0Å range:
|
Reference:
J.T.Henthorn,
R.J.Arias,
S.Koroidov,
T.Kroll,
D.Sokaras,
U.Bergmann,
D.C.Rees,
S.Debeer.
Localized Electronic Structure of Nitrogenase Femoco Revealed By Selenium K-Edge High Resolution X-Ray Absorption Spectroscopy. J.Am.Chem.Soc. V. 141 13676 2019.
ISSN: ESSN 1520-5126
PubMed: 31356071
DOI: 10.1021/JACS.9B06988
Page generated: Wed Jul 9 16:36:31 2025
ISSN: ESSN 1520-5126
PubMed: 31356071
DOI: 10.1021/JACS.9B06988
Last articles
F in 4ISFF in 4IKS
F in 4ISE
F in 4IQV
F in 4IQW
F in 4IQT
F in 4IQU
F in 4INB
F in 4IKT
F in 4IJU