Calcium »
PDB 6guz-6hhz »
6had »
Calcium in PDB 6had: Human Transketolase Variant E160Q
Enzymatic activity of Human Transketolase Variant E160Q
All present enzymatic activity of Human Transketolase Variant E160Q:
2.2.1.1;
2.2.1.1;
Protein crystallography data
The structure of Human Transketolase Variant E160Q, PDB code: 6had
was solved by
S.Dai,
V.Sautner,
K.Tittmann,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 42.75 / 1.04 |
| Space group | C 1 2 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 115.710, 85.490, 73.610, 90.00, 127.85, 90.00 |
| R / Rfree (%) | 11.1 / 13 |
Other elements in 6had:
The structure of Human Transketolase Variant E160Q also contains other interesting chemical elements:
| Magnesium | (Mg) | 1 atom |
| Sodium | (Na) | 1 atom |
Calcium Binding Sites:
The binding sites of Calcium atom in the Human Transketolase Variant E160Q
(pdb code 6had). This binding sites where shown within
5.0 Angstroms radius around Calcium atom.
In total only one binding site of Calcium was determined in the Human Transketolase Variant E160Q, PDB code: 6had:
In total only one binding site of Calcium was determined in the Human Transketolase Variant E160Q, PDB code: 6had:
Calcium binding site 1 out of 1 in 6had
Go back to
Calcium binding site 1 out
of 1 in the Human Transketolase Variant E160Q
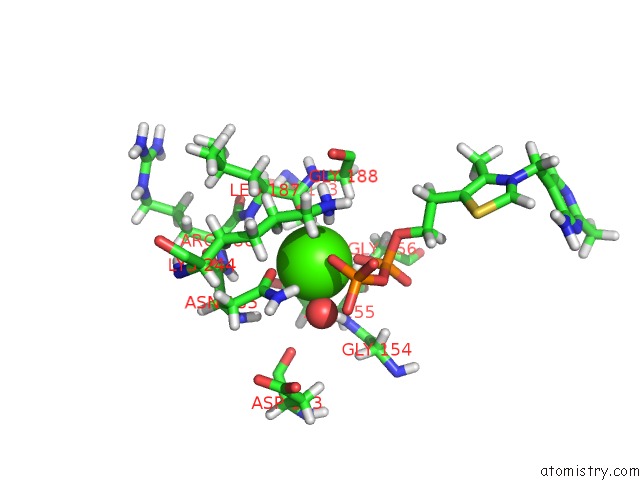
Mono view
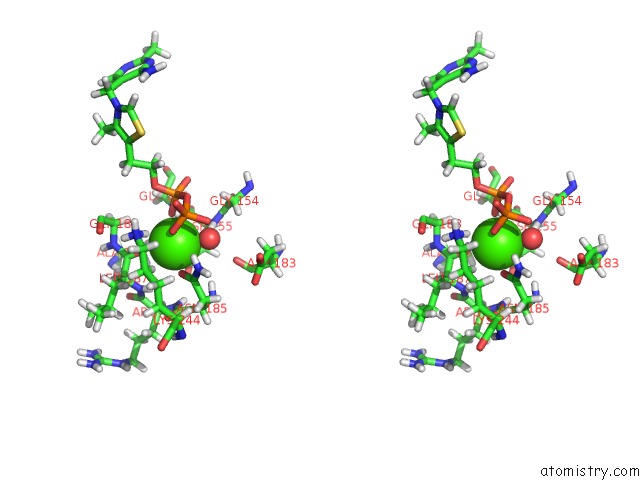
Stereo pair view

Mono view

Stereo pair view
A full contact list of Calcium with other atoms in the Ca binding
site number 1 of Human Transketolase Variant E160Q within 5.0Å range:
|
Reference:
S.Dai,
L.M.Funk,
F.R.Von Pappenheim,
V.Sautner,
M.Paulikat,
B.Schroder,
J.Uranga,
R.A.Mata,
K.Tittmann.
Low-Barrier Hydrogen Bonds in Enzyme Cooperativity. Nature V. 573 609 2019.
ISSN: ESSN 1476-4687
PubMed: 31534226
DOI: 10.1038/S41586-019-1581-9
Page generated: Wed Jul 9 14:33:38 2025
ISSN: ESSN 1476-4687
PubMed: 31534226
DOI: 10.1038/S41586-019-1581-9
Last articles
Ca in 7JUCCa in 7JUB
Ca in 7JTY
Ca in 7JU3
Ca in 7JOF
Ca in 7JRM
Ca in 7JRF
Ca in 7JRL
Ca in 7JRH
Ca in 7JNP